![SOLVED: Salt of a Strong Acid and a Weak Base pKa log [S] pH Salt of a Strong Base and a Weak Acid pKa pKw Jog [S] pH Salt of a Weak SOLVED: Salt of a Strong Acid and a Weak Base pKa log [S] pH Salt of a Strong Base and a Weak Acid pKa pKw Jog [S] pH Salt of a Weak](https://cdn.numerade.com/ask_images/21c70eb322cb4658bb80b94de9a1c863.jpg)
SOLVED: Salt of a Strong Acid and a Weak Base pKa log [S] pH Salt of a Strong Base and a Weak Acid pKa pKw Jog [S] pH Salt of a Weak

Relation between pH and log ( A − A )/( A − A ), the intercept (11.072)... | Download Scientific Diagram

Yexel Exams - Buffer solutions are used to reduce pH fluctuations associated with introduction of small amounts of strong acids or bases. Typical buffer solutions are composed of a weak acid or

BT_GS 1.9 Describe factors influencing the distribution of drugs (for example …. pH, pKa) …. | Primary LO of the Day
![The pH of basic buffer mixtures is given by : pH=pK(a)+log((["Base"])/(["Salt"])) , whereas pH of acidic buffer mixtures is given by: pH= pK(a)+log ((["Salt"])/(["Acid"])). Addition of little acid or base although shows no The pH of basic buffer mixtures is given by : pH=pK(a)+log((["Base"])/(["Salt"])) , whereas pH of acidic buffer mixtures is given by: pH= pK(a)+log ((["Salt"])/(["Acid"])). Addition of little acid or base although shows no](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/12003548_web.png)
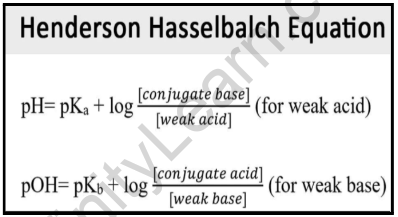
The pH of basic buffer mixtures is given by : pH=pK(a)+log((["Base"])/(["Salt"])) , whereas pH of acidic buffer mixtures is given by: pH= pK(a)+log ((["Salt"])/(["Acid"])). Addition of little acid or base although shows no
![SOLVED: Henderson-Hasselbalch Equation (YOu must be able to derive this equation): [conjugate base] [A-] pH pKa + log OR pH = pKa + log [acid] [HA] 0. 1. What is the pH SOLVED: Henderson-Hasselbalch Equation (YOu must be able to derive this equation): [conjugate base] [A-] pH pKa + log OR pH = pKa + log [acid] [HA] 0. 1. What is the pH](https://cdn.numerade.com/ask_images/b0b5d45010ed4627b06a3a093e4b025c.jpg)
SOLVED: Henderson-Hasselbalch Equation (YOu must be able to derive this equation): [conjugate base] [A-] pH pKa + log OR pH = pKa + log [acid] [HA] 0. 1. What is the pH
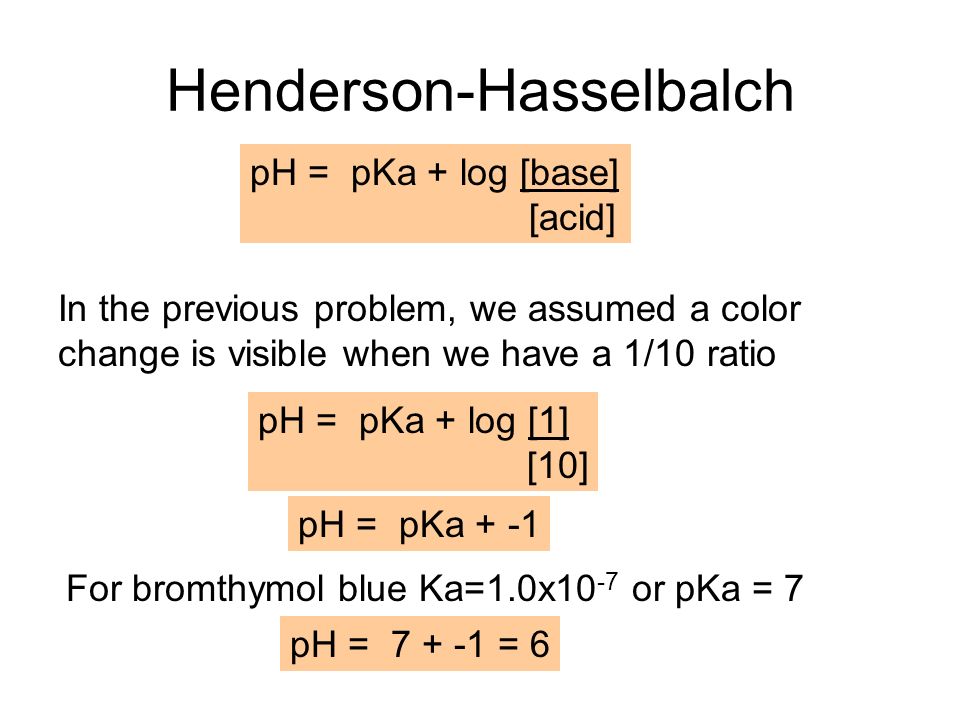


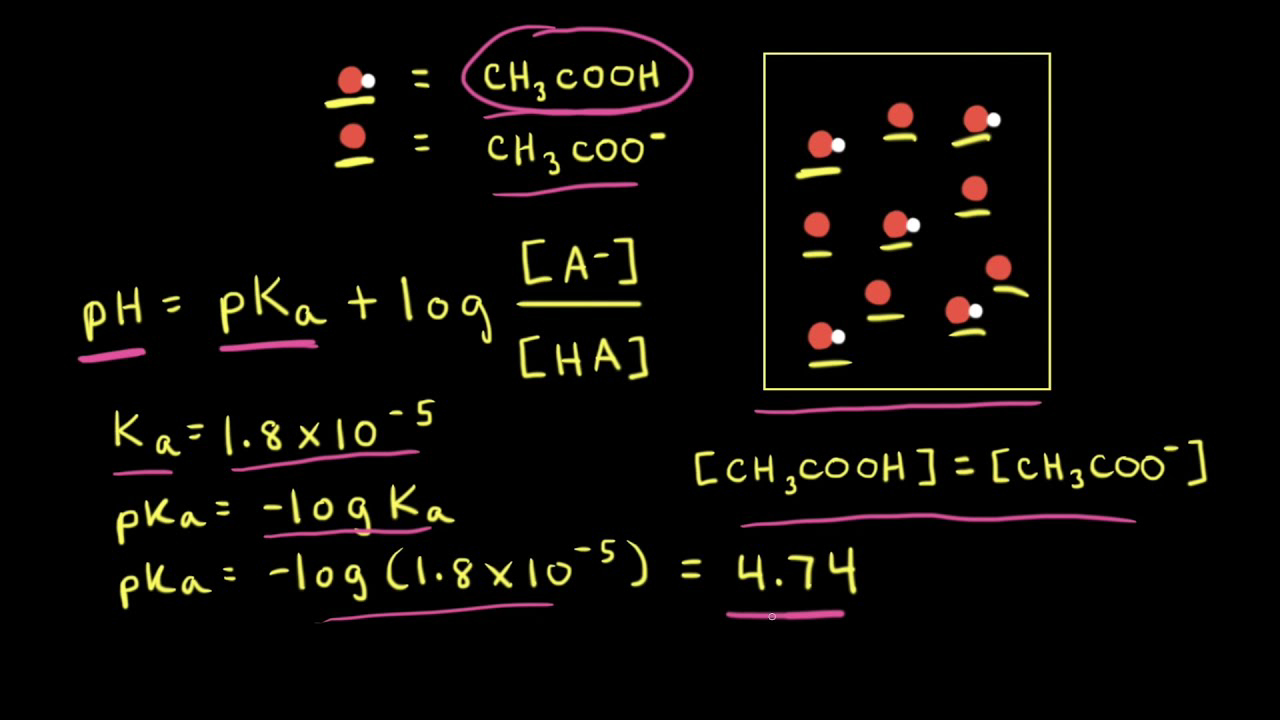
![Biochemistry | Henderson-Hasselbalch Equation Proof [pH=pKa] - YouTube Biochemistry | Henderson-Hasselbalch Equation Proof [pH=pKa] - YouTube](https://i.ytimg.com/vi/2jpB30LsT8g/maxresdefault.jpg)
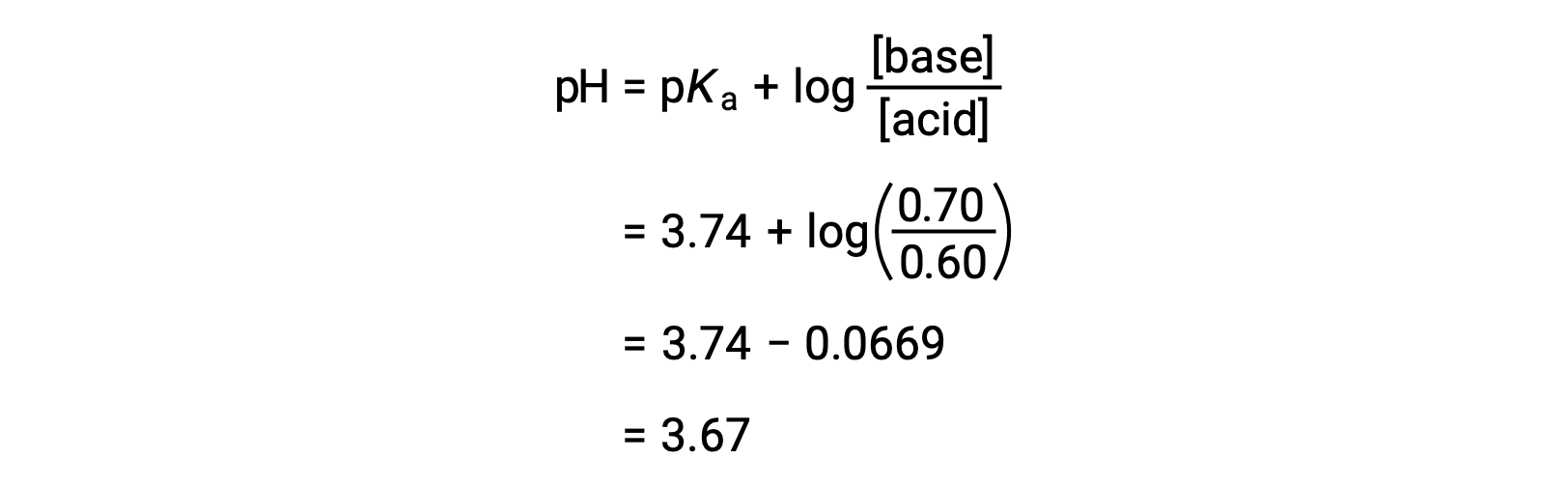

![SOLVED: [A-] log [HA] pKa pH d. [A- ] PH = PKa + log [HA] [A-] PH = pKa + [HA] SOLVED: [A-] log [HA] pKa pH d. [A- ] PH = PKa + log [HA] [A-] PH = pKa + [HA]](https://cdn.numerade.com/ask_images/959424b8320b49c898de609f834a314d.jpg)
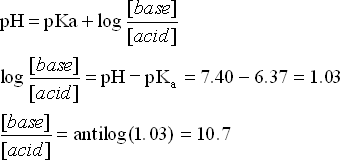









![Solved Just as pH is the negative logarithm of (H30+], pK, | Chegg.com Solved Just as pH is the negative logarithm of (H30+], pK, | Chegg.com](https://media.cheggcdn.com/media/375/3755951c-9b2e-4931-a914-33bb100113d8/php72x3Cn.png)